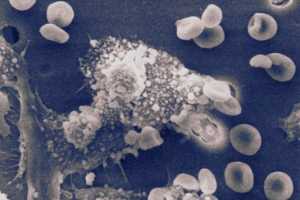
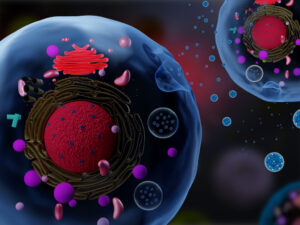
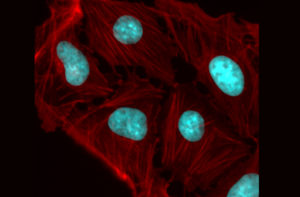
Fat is a normal and necessary part of the body. Fat cells store and release energy, as well as play significant roles in hormonal regulation and immunity.
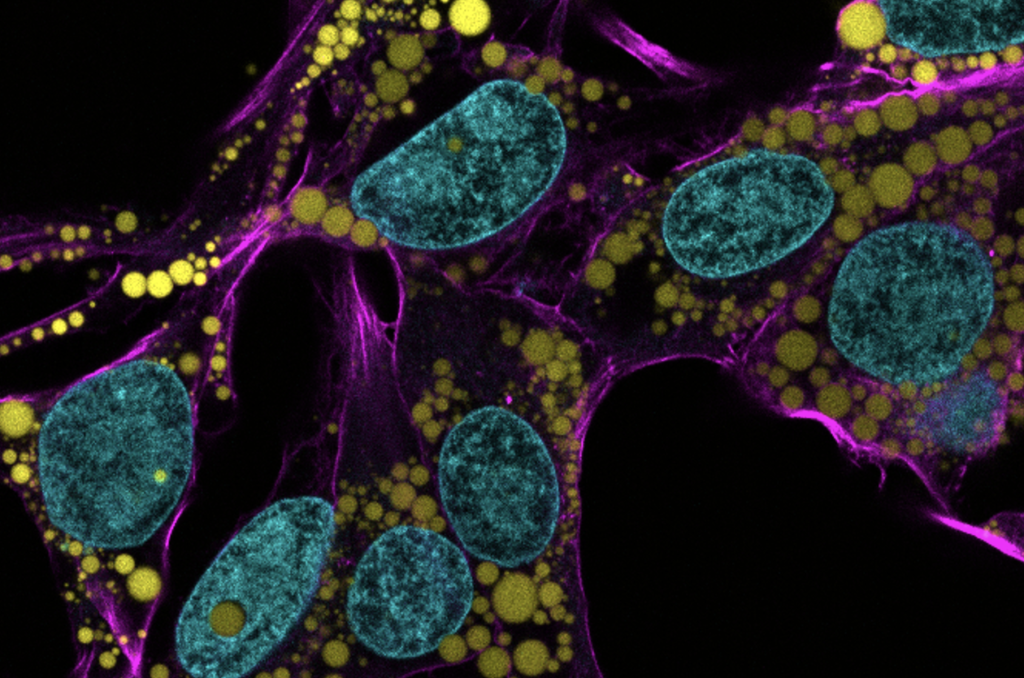
In recent decades, a concerning rise in metabolic illnesses – such as cardiovascular disease, high blood pressure and diabetes – has focused scientific attention on the biology and chemistry of fat, resulting in a wealth of information about how fat cells work.
But fat cells and their metabolic activities are only part of the story.
Fat-filled lipid droplets, tiny spheres of fat many times smaller than fat cells, are a growing subject of scientific interest. Found inside many different cell types, these lipid particles have long been little understood. Studies have begun to illuminate these droplets’ participation in metabolic functions and cellular protection, but we still know next to nothing about the physical nature of fat.
Now, researchers at the University of Pennsylvania School of Engineering and Applied Science have looked beyond biochemistry to publish groundbreaking work on the physics of these droplets, revealing them to be a potential threat to a cell’s nucleus. In the August issue of the Journal of Cell Biology, they are the first to discover fat-filled lipid droplets’ surprising capability to indent and puncture the nucleus, the organelle which contains and regulates a cell’s DNA.
The stakes of their findings are high: a ruptured nucleus can lead to elevated DNA damage that is characteristic of many diseases, including cancer.
The study was led by Dennis E. Discher, Robert D. Bent Professor in the Department of Chemical and Biomolecular Engineering, Bioengineering, and in Mechanical Engineering and Applied Mechanics, Irena Ivanovska, Ph.D. research associate in Penn’s Molecular and Cell Biophysics Lab, and Michael Tobin, Ph.D. candidate in the Department of Bioengineering.
“Intuitively, people think of fat as soft,” says Discher. “And on a cellular level it is. But at this small size of droplet— measuring just a few microns rather than the hundreds of microns of a mature fat cell—it stops being soft. Its shape has a much higher curvature, bending other objects very sharply. This changes its physics in the cell. It can deform. It can damage. It can rupture.”
Read the full story in Penn Engineering Today.