by Sophie Burkholder
Vector Flow Imaging Helps Visualize Blood Flow in Pediatric Hearts
 A group of biomedical engineers at the University of Arkansas used a new ultrasound-based imaging technique called vector flow imaging to help improve the diagnosis of congenital heart disease in pediatric patients. The study, led by associate professor of biomedical engineering Morten Jensen, Ph.D., collaborated with cardiologists at the local Children’s Hospital in Little Rock to produce images of the heart in infants to help potentially diagnose congenital heart defects. Though the use of vector flow imaging has yet to be developed for adult patients, this type of imaging could possibly provide more detail about the direction of blood flow through the heart than traditional techniques like echocardiography do. In the future, the use of both techniques could provide information about both the causes and larger effects of heart defects in patients.
A group of biomedical engineers at the University of Arkansas used a new ultrasound-based imaging technique called vector flow imaging to help improve the diagnosis of congenital heart disease in pediatric patients. The study, led by associate professor of biomedical engineering Morten Jensen, Ph.D., collaborated with cardiologists at the local Children’s Hospital in Little Rock to produce images of the heart in infants to help potentially diagnose congenital heart defects. Though the use of vector flow imaging has yet to be developed for adult patients, this type of imaging could possibly provide more detail about the direction of blood flow through the heart than traditional techniques like echocardiography do. In the future, the use of both techniques could provide information about both the causes and larger effects of heart defects in patients.
Using Stem Cells to Improve Fertility in Leukemia Survivors
One of the more common side effects of leukemia treatment in female patients is infertility, but researchers at the University of Michigan want to change that. Led by associate professor of biomedical engineering Ariella Shikanov, Ph.D., researchers in her lab found ways of increasing ovarian follicle productivity in mice, which directly relates to the development of mature eggs. The project involves the use of adipose-derived stem cells, that can be found in human fat tissue, to surround the follicles in an ovary-like, three-dimensional scaffold. Because the radiation treatments for leukemia and some other cancers are harmful to follicles, increasing their survival rate with this stem cell method could reduce the rate of infertility in patients undergoing these treatments. Furthermore, this new approach is innovative in its use of a three-dimensional scaffold as opposed to a two-dimensional one, as it stimulates follicle growth in all directions and thus helps to increase the follicle survival rate.
Penn Engineers Look at How Stretching & Alignment of Collagen Fibers Help Cancer Cells Spread
Cancer has such a massive impact on people’s lives that it might be easy to forget that the disease originates at the cellular level. To spread and cause significant damage, individual cancer cells must navigate the fibrous extracellular environment that cells live in, an environment that Penn Engineer Vivek Shenoy has been investigating for years.
Shenoy is the Eduardo D. Glandt President’s Distinguished Professor with appointments in Materials Science and Engineering, Mechanical Engineering and Applied Mechanics, and Bioengineering. He is also the Director of the Center for Engineering MechanoBiology (CEMB), one of the NSF’s twelve Science and Technology Centers.
Shenoy’s most recent study on cancer’s mechanical environment was led by a postdoctoral researcher in his lab, Ehsan Ban. Paul Janmey, professor in Physiology and Bioengineering, and colleagues at Stanford University also contributed to the study. Shenoy also received the Heilmeier Award this March and delivered the Heilmeier Award Lecture in April.
Read the rest of this story on Penn Engineering’s Medium Blog.
Controlled Electrical Stimulation Can Prevent Joint Replacement Infections
Joint replacements are one of the most common kinds of surgery today, but they still require intense post-operative therapy and have a risk of infection from the replacement implant. These infections are usually due to the inflammatory response that the body has to any foreign object, and can become serious and life-threatening if left untreated. Researchers at the University of Buffalo Jacobs School of Medicine and Biomedical Sciences hope to offer a solution to preventing infections through the use of controlled electrical stimulation. Led by Mark Ehrensberger, Ph.D., Kenneth A. Krackow, M.D., and Anthony A. Campagnari, Ph.D., the treatment system uses the electrical signal to create an antibacterial environment at the interface of the body and the implant. While the signal does not prevent infections completely, these antibacterial properties will prevent infections from worsening to a more serious level. Patented as the Biofilm Disruption Device TM, the final product uses two electrode skin patches and a minimally invasive probe that delivers the electrical signal directly to the joint-body interface. The researchers behind the design hope that it can help create a more standard way of effectively treating joint replacement infections.
People and Places

For their senior design project, four bioengineering seniors — Gabriel Koo, Ethan Zhao, Daphne Cheung, and Shelly Teng — created a low-cost tuberculosis diagnostic that they called TBx. Using their knowledge of the photoacoustic effect of certain dyes, the platform the group created can detect the presence of lipoarabinomannan in patient urine. The four seniors presented TBx at the Rice360 Design Competition in Houston, Texas this spring, which annually features student-designed low-cost global health technologies.

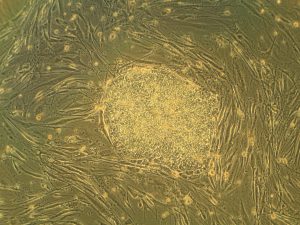 Heart attacks are the result of a stoppage of blood flow to the heart – an interruption to normal function that can result in severe tissue damage, or even tissue death. This loss of healthy tissue function is one of the biggest challenges in treating patients that undergo heart attacks, as the damaged tissue increases their risk of having future attacks. One of the main solutions to this issue right now is the creation of cardiac tissue scaffolds using stem cells to create a platform for new and healthy tissue to grow in vivo. A group of biomedical engineers at Michigan Technological University hopes to
Heart attacks are the result of a stoppage of blood flow to the heart – an interruption to normal function that can result in severe tissue damage, or even tissue death. This loss of healthy tissue function is one of the biggest challenges in treating patients that undergo heart attacks, as the damaged tissue increases their risk of having future attacks. One of the main solutions to this issue right now is the creation of cardiac tissue scaffolds using stem cells to create a platform for new and healthy tissue to grow in vivo. A group of biomedical engineers at Michigan Technological University hopes to 